THYMOSIN BETA-4 (43aa) - 10mg
Our Commitment to Analytical Transparency
Whenever available, our third-party Certificates of Analysis (COAs) include:
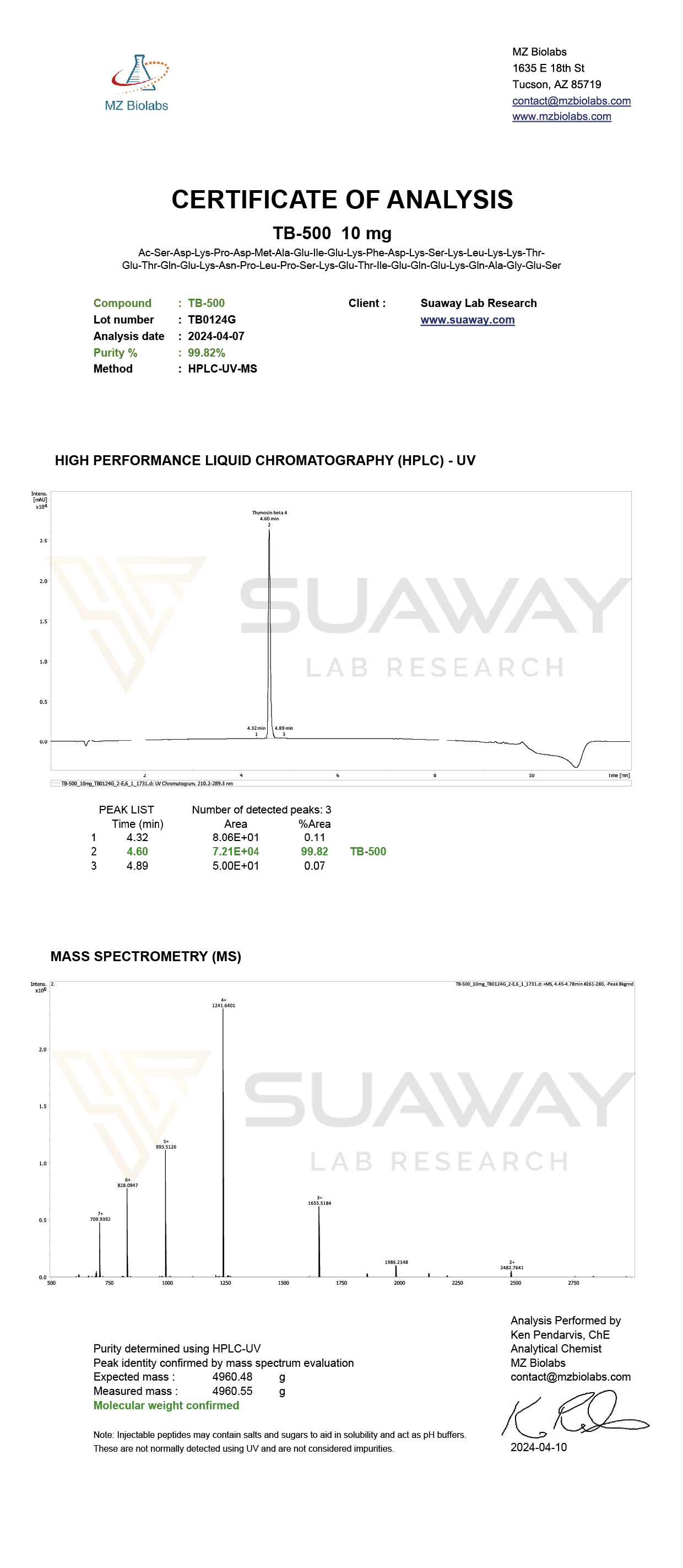
- HPLC purity testing
- Mass Spectrometry (MS) identity confirmation
- Net Peptide Content (NPC) analysis
We believe that Net Peptide Content analysis is particularly important for evaluating the actual amount of peptide present, taking into account residual moisture, counterions, and other non-peptide components.
Thymosin Beta-4 (Tβ4), often associated with the peptide fragment TB-500 in research contexts, is a naturally occurring peptide involved in tissue repair, immune regulation, and cellular regeneration. Its primary mechanism is linked to actin-binding activity, supporting cytoskeletal remodeling and promoting cell migration essential for wound healing. Research suggests Tβ4 may stimulate angiogenesis, enhance collagen deposition, support keratinocyte and endothelial migration, and reduce inflammatory cytokine signaling through modulation of pathways such as NF-κB. It has been studied in experimental models of skin repair, corneal injury, dry eye conditions, cardiovascular remodeling after ischemic injury, systemic inflammation including sepsis models, neurological injury, autoimmune neuroinflammation, and musculoskeletal recovery. These properties make Tβ4 a major peptide of interest in regenerative biology and inflammation-related research.
Description
SPECIFICATIONS
Product Code: TB4010
Sequence: Ac-Ser-Asp-Lys-Pro-Asp-Met-Ala-Glu-Ile-Glu-Lys-Phe-Asp-Lys-Ser-Lys-Leu-Lys-Lys-Thr-Glu-Thr-Gln-Glu-Lys-Asn-Pro-Leu-Pro-Ser-Lys-Glu-Thr-Ile-Glu-Gln-Glu-Lys-Gln-Ala-Gly-Glu-Ser
Molecular Formula: C212H350N56O78S
Molecular Weight: 4960.55 g/mol
CAS: 77591-33-4
Purity: Technical / Research Grade ≥98%
Other details: No TFA Salt
Form: Lyophilized powder
Color: White
Storage temperature: -20°C
Source: Synthetic
Safety classification: Standard handling
DESCRIPTION
Thymosin Beta-4 (Tβ4), often associated in research contexts with the peptide fragment commonly referred to as TB-500, is a naturally occurring peptide that has attracted considerable scientific interest due to its broad role in tissue repair, immune regulation, and cellular regeneration. Thymosins are peptides originally associated with thymus activity and immune development, and they are widely studied for their involvement in biological signaling pathways that influence inflammation control, wound healing, and cellular migration.
Tβ4 is present in many tissues throughout the body and is known to become upregulated in response to injury. It is produced by platelets and various cell types during tissue damage, suggesting that it functions as an endogenous repair peptide involved in limiting injury expansion and promoting recovery processes. Because tissue regeneration requires coordinated vascular growth, extracellular matrix remodeling, and immune balance, Tβ4 has become a peptide of major interest in regenerative biology and inflammation-related research.
Biological role and physiological relevance
The immune system relies on highly coordinated signaling molecules to maintain balance between defense and inflammation. The thymus is known as a key organ involved in immune development, particularly in supporting the maturation of T lymphocytes (T cells), which play a central role in adaptive immunity. Thymosin-related peptides are traditionally associated with immune modulation, supporting immune system maturation and function.
In addition to immune regulation, Tβ4 is widely recognized as a peptide involved in tissue protection and regeneration. Research suggests that it participates in cellular repair processes by supporting cell survival, reducing excessive inflammatory signaling, and enhancing regeneration mechanisms following injury. Because inflammation is a common pathological driver in numerous diseases, peptides capable of modulating inflammatory cascades are increasingly studied for broad systemic applications.
Mechanism of action: actin regulation and cellular migration
One of the most important mechanisms associated with Thymosin Beta-4 is its interaction with actin, a fundamental structural protein of the cytoskeleton. Actin is essential for cell movement, shape regulation, and tissue remodeling. Tβ4 is considered a major actin-binding peptide and is believed to regulate actin polymerization dynamics, thereby influencing cell migration and cellular structural organization.
This mechanism is highly relevant to wound repair, because healing requires the migration of keratinocytes, endothelial cells, fibroblasts, and immune cells into the damaged area. By supporting actin-dependent cellular mobility, Tβ4 may contribute to accelerated wound closure, tissue remodeling, and regeneration of damaged structures.
Research has suggested that Tβ4 is among the earliest genes activated following tissue injury, highlighting its potential role as an early repair mediator. Once induced, the peptide appears to promote a repair cascade involving angiogenesis, extracellular matrix remodeling, and reduction of inflammatory mediators.
Angiogenesis and tissue remodeling
Tβ4 has been studied extensively for its potential ability to support angiogenesis, the formation of new blood vessels from existing vascular structures. Angiogenesis is essential for tissue healing because injured tissue requires increased blood supply to deliver oxygen, nutrients, immune mediators, and repair signals.
Experimental studies suggest that Tβ4 may stimulate endothelial cell differentiation and migration, supporting microvascular regeneration. This effect is considered important in healing processes involving skin injuries, corneal damage, muscle trauma, and ischemic tissues. Increased vascularization may also support faster recovery of connective tissue structures such as tendons and ligaments.
Tβ4 has also been studied for its ability to influence collagen deposition and extracellular matrix remodeling. Proper collagen organization is essential for tissue integrity, flexibility, and structural repair. The peptide has been associated with enhanced keratinocyte migration and improved wound closure, indicating that its activity extends beyond vascular growth into structural reconstruction of damaged tissues.
Anti-inflammatory and immune-modulating properties
A major reason why Tβ4 has become widely studied is its association with inflammation control. Chronic or excessive inflammation can delay healing and contribute to tissue degeneration. Research suggests that Tβ4 may reduce inflammatory signaling by modulating cytokine production and influencing key inflammatory pathways.
In experimental contexts, Tβ4 has been described as influencing transcription factors such as NF-κB, which regulates numerous inflammatory genes. By modulating inflammatory cascades, Tβ4 may reduce the production of inflammatory cytokines such as TNF-related mediators and may influence matrix metalloproteinases (MMPs), enzymes involved in extracellular matrix breakdown. This is significant because excessive MMP activity is often associated with tissue destruction and chronic inflammatory pathology.
Because inflammation is a major contributor in conditions such as autoimmune disorders, chronic fatigue-related syndromes, infections, and degenerative joint conditions, the anti-inflammatory profile of Tβ4 has led to research interest in its systemic biological relevance.
Skin repair and wound healing research
Tβ4 has been investigated in multiple experimental models of skin injury. Research involving dermal wound healing has suggested that topical or localized administration of Tβ4 can accelerate healing processes by supporting collagen deposition, keratinocyte migration, and angiogenesis.
In animal studies involving full-thickness skin wounds, Tβ4 treatment has been associated with improved repair rates compared to controls. These effects were also observed in models of impaired wound healing, such as aging-related delayed repair and diabetic wound models, where tissue regeneration capacity is reduced.
Such findings have contributed to the scientific view that Tβ4 functions as an endogenous wound repair peptide capable of accelerating structural recovery in tissues with compromised healing ability.
Corneal repair and ocular surface research
The cornea is a highly sensitive tissue where wound healing is strongly influenced by inflammation and epithelial migration. Corneal burns and chemical injuries often cause intense inflammation and may lead to long-term visual impairment.
Experimental studies suggest that Tβ4 may support corneal epithelial regeneration by enhancing epithelial cell migration across damaged corneal surfaces. Research has also described reduced inflammatory signaling and increased expression of repair-associated proteins such as laminin-5, which supports epithelial adhesion and migration. In experimental models, Tβ4 has been associated with reduced apoptosis and enhanced angiogenic response during corneal recovery.
These findings have positioned Tβ4 as a peptide of interest in ocular surface research, including dry eye-related conditions and corneal injury recovery models.
Cardiovascular repair and cardioprotective research
Tβ4 has also been investigated in cardiovascular research due to its potential effects on tissue remodeling after cardiac injury. Following ischemic damage, such as myocardial infarction, the heart undergoes remodeling processes that can lead to fibrosis and reduced contractile function.
In experimental models, Tβ4 has been described as cardioprotective, supporting cell survival, reducing fibrotic remodeling, and enhancing migration of repair-associated cells. Some research suggests that Tβ4 may activate pathways such as integrin-linked kinase signaling, which is involved in cellular survival, migration, and tissue repair responses.
These effects have made Tβ4 a peptide of interest in studies focused on reducing infarct-related damage, supporting cardiac function recovery, and limiting pathological scar formation.
Sepsis and systemic inflammation models
Septic shock is characterized by extreme inflammatory activation, cytokine imbalance, and systemic tissue injury. Because pro-inflammatory cytokines play a major role in septic pathology, peptides that reduce cytokine release have been explored in experimental settings.
Research has suggested that Tβ4 may reduce inflammatory mediator levels and decrease lethality in endotoxin-induced sepsis models. Interestingly, experimental observations have also reported that endogenous Tβ4 levels may decrease during induced sepsis, supporting the hypothesis that Tβ4 could be a protective endogenous factor whose depletion worsens inflammatory outcomes.
These findings have contributed to interest in Tβ4 as a peptide involved in systemic inflammation regulation and immune resilience research.
Neurological protection and nerve recovery research
The nervous system is particularly vulnerable to inflammatory stress, oxidative injury, and excitotoxic damage. Tβ4 has been investigated in neurological models for its potential protective effects on neurons and supporting cells.
Experimental research suggests that Tβ4 may reduce neuronal loss in models involving hippocampal injury and excitotoxic stress. It has also been studied in autoimmune encephalomyelitis models, where treatment was associated with reduced inflammation, increased numbers of mature oligodendrocytes, and improved functional outcomes. These findings indicate potential relevance in research involving demyelination, neuroinflammation, and nerve regeneration pathways.
Tβ4 has also been reported to protect neuronal cells in vitro against ethanol toxicity and glutamate-induced toxicity, further supporting its role in neuronal resilience research.
Musculoskeletal recovery and connective tissue support
Because actin regulation is central to cellular migration and tissue remodeling, Tβ4 has become a peptide of interest in musculoskeletal repair research. Studies suggest that it may support muscle fiber recovery, reduce inflammation-related stiffness, and improve structural remodeling of tendons, ligaments, and connective tissue.
It has also been discussed for its potential role in preventing formation of adhesions and fibrotic bands, which can impair flexibility and movement. These properties have led to interest in Tβ4 in contexts related to recovery from muscle injury, connective tissue strain, and chronic musculoskeletal inflammation.
Additional research observations
Beyond tissue healing, some research discussions have suggested additional systemic effects such as improved flexibility, reduced muscular cramping, enhanced tissue regeneration capacity, and possible effects on hair follicle activity. While these areas remain less established compared to wound healing and tissue repair models, they reflect the broad biological influence of Tβ4-related pathways.
Research perspective
Thymosin Beta-4 is widely regarded as a multifunctional peptide involved in repair signaling, angiogenesis, cytoskeletal remodeling, and inflammation regulation. Its role as an actin-binding peptide places it at the center of cellular migration and tissue regeneration biology, while its anti-inflammatory signaling profile makes it relevant to systemic disease models where chronic inflammation contributes to degeneration.
Due to its broad biological activity, Tβ4 remains a peptide of high interest in regenerative medicine research, cardiovascular recovery models, ocular surface healing studies, inflammatory disease research, and neuroprotective investigations.
REFERENCES
All information presented above is derived from in vitro experiments, animal studies, and other preclinical research models. These data are intended solely for basic scientific investigation of biological mechanisms and do not imply any therapeutic, diagnostic, preventive, or clinical use in humans or animals.
A. Goldstein et al., "Thymosin β4: a multi-functional regenerative peptide. Basic properties and clinical applications" [PubMed]
A. Goldstein et al., "Advances in the basic and clinical applications of thymosin β4" [PubMed]
D. Crockford et al., "Thymosin beta4: structure, function, and biological properties supporting current and future clinical applications" [PubMed]
G. Sosne et al., "Biological activities of thymosin beta4 defined by active sites in short peptide sequences" [PubMed]
E.G. Yarmola et al., "Thymosin beta4: actin regulation and more" [PubMed]
M. Badamchian et al., "Thymosin beta(4) reduces lethality and down-regulates inflammatory mediators in endotoxin-induced septic shock" [PubMed]
A. Gonzales-Franquesa et al., "Discovery of thymosin β4 as a human exerkine and growth factor" [PubMed]
J.D. Young et al., "Thymosin beta 4 sulfoxide is an anti-inflammatory agent generated by monocytes in the presence of glucocorticoids" [PubMed]
M.R. Bubb "Thymosin beta 4 interactions" [PubMed]
Y. Xing et al., "Progress on the Function and Application of Thymosin β4" [PubMed]
N. Shomali et al., "A new insight into thymosin β4, a promising therapeutic approach for neurodegenerative disorders" [PubMed]
S. Munshaw et al., "Thymosin β4 protects against aortic aneurysm via endocytic regulation of growth factor signaling" [PubMed]
G. Renga et al., "Thymosin β4 limits inflammation through autophagy" [PubMed]
Y. Wang et al., "Adjunctive Thymosin Beta-4 Treatment Influences MΦ Effector Cell Function to Improve Disease Outcome in Pseudomonas aeruginosa-Induced Keratitis" [PubMed]
K.W. Freeman et al., "Regenerative protein thymosin beta-4 is a novel regulator of purinergic signaling" [PubMed]
G. Sosne et al., "Thymosin beta 4 promotes corneal wound healing and decreases inflammation in vivo following alkali injury" [PubMed]
J. Zhang et al., "Thymosin beta4 promotes oligodendrogenesis in the demyelinating central nervous system" [PubMed]
Rui Yu et al., "Recombinant Human Thymosin Beta-4 Protects against Mouse Coronavirus Infection" [PubMed]
DISCLAIMER
All information This product is intendend for lab research and development use only. These studies are performed outside of the body. This product is not medicines or drugs and has not been approved by the FDA or EMA to prevent, treat or cure any medical condition, ailment or disease. Bodily introduction of any kind into humans or animals is strictly forbidden by law. This product should only be handled by licensed, qualified professionals.
All product information provided on this website is for informational and educational purposes only.
INDEPENDENT THIRD-PARTY CERTIFICATE OF ANALYSIS SHOWN FOR REFERENCE PURPOSES. ANALYTICAL RESULTS MAY VARY SLIGHTLY BETWEEN BATCHES.
Data sheet
Customers who bought this product also bought:
| Image | Ref. | Unit price | Weight | Avail. | Buy |
|---|---|---|---|---|---|
 | TB4010 | €120.00 Discount by quantity 10+ €96.00 –20% 20+ €84.00 –30% 50+ €74.40 –38% 100+ €67.20 –44% 200+ €60.00 –50% | 0.000000 kg |  | |
| Total: 0,0 All prices with tax included. | |||||
 Product is available
Product is available Product is no longer in stock
Product is no longer in stock Product is no in stock, but is allowed to be back-ordered
Product is no in stock, but is allowed to be back-ordered