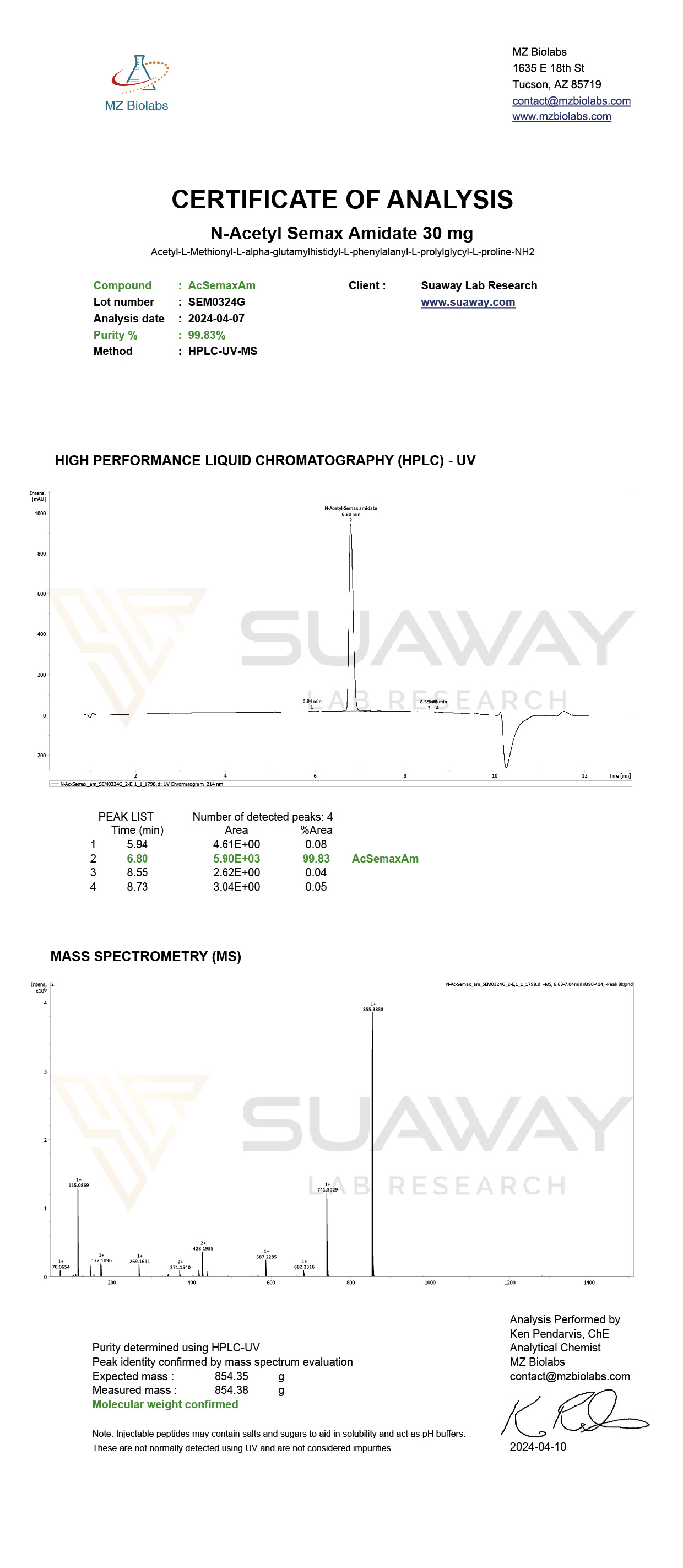
N-ACETYL SEMAX AMIDATE - 30mg
N-Acetyl Semax Amidate is a synthetic peptide that has been widely investigated in experimental research for its involvement in neurogenic and neurorestorative biological processes. Scientific studies have explored its activity in relation to central nervous system signaling, cognitive performance, vascular-associated mechanisms, and immune-related pathways. Research has primarily examined N-Acetyl Semax Amidate in the context of the following biological effects:
Neuroprotective pathways associated with neuronal resilience
Mechanisms involved in limiting nerve damage and supporting neural integrity
Cognitive functions related to memory, learning, and information processing
Cardiovascular-related signaling studied in ischemic and hypoxic research models
Neurobehavioral pathways relevant to attention regulation
Modulation of attention and focus-related neural activity
Stress- and pain-associated signaling pathways explored in experimental settings
Description
SPECIFICATIONS
Product Code: NSX030
Sequence: Ac-Met-Glu-His-Phe-Pro-Gly-Pro-NH₂
Molecular Formula: C39H54N10O10S
Molecular Weight: 854.38 g/mol
CAS: 80714-61-0
Purity: Technical / Research Grade ≥99%
Other details: No TFA Salt
Form: Lyophilized powder
Color: White
Storage temperature: -20°C
Source: Synthetic
Safety classification: Standard handling
DESCRIPTION
Adrenocorticotropic hormone fragment 4–10 is a naturally occurring fragment of human adrenocorticotropic hormone (ACTH) and is structurally analogous to the synthetic polypeptide N-Acetyl Semax Amidate (ACTH 4–10). Unlike full-length ACTH, this fragment does not exhibit classical endocrine effects but instead displays distinct activity within the central nervous system, where it has been studied for its influence on attention, memory, and cognitive performance.
Experimental research indicates that N-Acetyl Semax Amidate exhibits psychostimulant-like properties, including increased dopamine synthesis and release, as well as elevated levels of brain-derived neurotrophic factor (BDNF). These molecular effects are associated with enhanced attentional capacity and support of neuroplastic mechanisms involved in neural adaptation.
Studies in animal models have examined changes in 5-hydroxyindoleacetic acid (5-HIAA), the primary metabolite of serotonin, following peptide administration. Increased 5-HIAA levels were observed within hours, suggesting modulation of monoaminergic neurotransmission.
Research suggests that N-Acetyl Semax Amidate enhances dopaminergic activity in the striatum, a brain region involved in cognition, motivation, and motor coordination.
Clinical investigations conducted in Russia evaluated intranasal Semax administration in healthy volunteers under stress conditions. Participants receiving doses ranging from 0.015 to 0.050 mg/kg demonstrated improvements in attention and memory measures without reported adverse effects in controlled settings.
Additional research has examined Semax in experimental models of acute cerebral hypoxia and ischemic stroke. Studies in rodents indicate modulation of gene expression related to vascular function, erythropoiesis, smooth muscle migration, and angiogenesis.
Gene-regulatory effects may contribute to neuroprotective observations in experimental stroke models, potentially through enhanced nutrient delivery, neuronal support, mitochondrial stabilization, and improved cellular energy dynamics.
In healthy animal models, intranasal administration has been associated with altered gene expression in the hippocampus and frontal cortex within minutes, including increased expression of nerve growth factor (NGF) and BDNF.
Research exploring neurotrophic signaling suggests that peptides enhancing BDNF expression may influence pathways relevant to cognitive adaptability and mood regulation.
Semax has also been evaluated in animal models of myocardial infarction. Administration at 150 µg/kg for six consecutive days post-infarction was associated with modulation of ventricular remodeling and prevention of increases in left ventricular diastolic pressure.
Peptide Modifications: N-Acetylation and Amidation
N-acetylation is a modification used in synthetic chemistry to alter peptide charge, hydrophobicity, and stability. This modification can influence peptide half-life, tissue localization, and receptor interaction while generally preserving core biological activity.
Amidation affects the C-terminal region of peptides and reduces susceptibility to proteolytic degradation. It may enhance receptor binding affinity, extend circulatory persistence, and improve resistance to pH fluctuations, contributing to more stable biological activity.
REFERENCES
All observations described above originate from in vitro systems, animal studies, or other preclinical experimental models. They are intended solely to support basic research into molecular, cellular, and physiological mechanisms and do not imply therapeutic, diagnostic, or preventive applications in humans or animals.
T. L. Agapova et al., “Effect of semax on the temporary dynamics of brain-derived neurotrophic factor and nerve growth factor gene expression in the rat hippocampus and frontal cortex” [PubMed]
I. S. Lebedeva et al., “Effects of Semax on the Default Mode Network of the Brain” [PubMed]
K. O. Eremin et al., “Semax, an ACTH(4-10) analogue with nootropic properties, activates dopaminergic and serotoninergic brain systems in rodents” [PubMed]
E. I. Gusev et al., “The efficacy of semax in the treatment of patients at different stages of ischemic stroke” [PubMed]
Shih-Jen Tsai, “Semax, an analogue of adrenocorticotropin (4-10), is a potential agent for the treatment of attention-deficit hyperactivity disorder and Rett syndrome” [PubMed]
E. I. Gusev et al., “Effectiveness of semax in acute period of hemispheric ischemic stroke” [PubMed]
S. I. Gusev et al., “Semax in prevention of disease progress and development of exacerbations in patients with cerebrovascular insufficiency” [PubMed]
O. V. Dolotov et al., “Semax binds specifically and increases levels of brain-derived neurotrophic factor protein in rat basal forebrain” [PubMed]
V. Stavchansky et al., “Effect of Semax and its C-end peptide PGP on morphology and proliferative activity of rat brain cells during experimental ischemia” [PubMed]
L. V. Dergunova et al., “The Peptide Drug ACTH(4-7)PGP (Semax) Suppresses mRNA Transcripts Encoding Proinflammatory Mediators Induced by Reversible Ischemia of the Rat Brain” [PubMed]
A. Magrì et al., “Influence of the N-terminus acetylation of Semax on copper(II) and zinc(II) coordination and biological properties” [PubMed]
M. Shypshyna et al., “Effect of peptide semax on synaptic activity and short-term plasticity of glutamatergic synapses” [PubMed]
E. V. Iakovleva et al., “Study of the efficacy of semax in global cerebral ischemia in vivo” [PubMed]
DISCLAIMER
This product is intended for laboratory research and development use only. These studies are performed outside of the body. This product is not a medicine or drug and has not been approved by the FDA or EMA to prevent, treat, or cure any medical condition, ailment, or disease.
Bodily introduction of any kind into humans or animals is strictly forbidden by law. This product should only be handled by licensed, qualified professionals.
All product information provided on this website is for informational and educational purposes only.
INDEPENDENT THIRD-PARTY CERTIFICATE OF ANALYSIS SHOWN FOR REFERENCE PURPOSES. ANALYTICAL RESULTS MAY VARY SLIGHTLY BETWEEN BATCHES.
Data sheet
Customers who bought this product also bought:
| Image | Ref. | Unit price | Weight | Avail. | Buy |
|---|---|---|---|---|---|
 | NSX030 | €75.00 Discount by quantity 10+ €63.75 –15% 20+ €54.00 –28% 50+ €45.00 –40% 100+ €39.00 –48% 200+ €33.75 –55% | 0.000000 kg |  | |
| Total: 0,0 All prices with tax included. | |||||
 Product is available
Product is available Product is no longer in stock
Product is no longer in stock Product is no in stock, but is allowed to be back-ordered
Product is no in stock, but is allowed to be back-ordered










